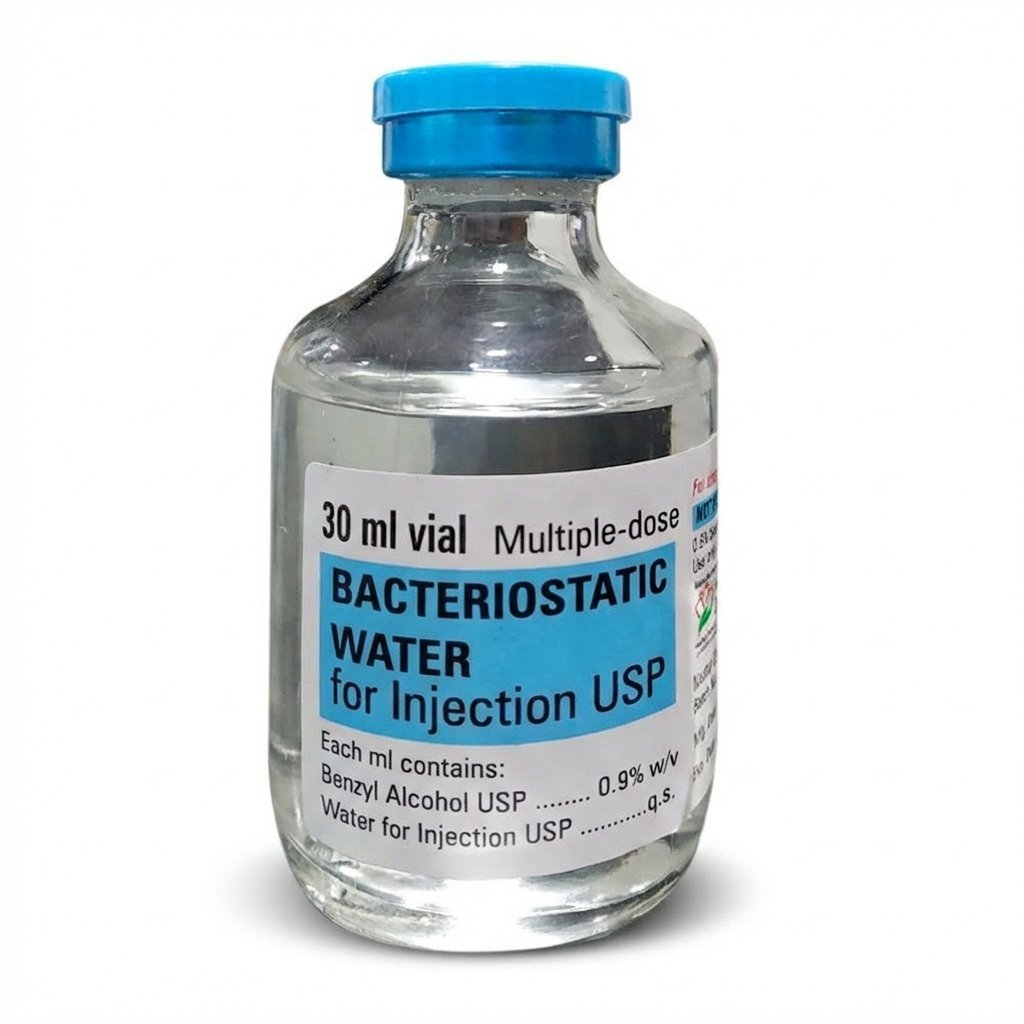
Bacteriostatic Water for Injection 30mL
€25.00
Product Specifications
| Specification | Detail |
|---|---|
| Product Name | Bacteriostatic Water for Injection 30mL |
| Composition | 99.1% WFI Water, 0.9% Benzyl Alcohol |
| Preservative | 0.9% (9 mg/mL) benzyl alcohol |
| Form | Liquid, ready-to-use solution |
| Fill Volume | 30 mL multi-dose vial |
| Sterilization | 0.22 µm filtration, aseptically filled |
| pH Range | 4.5–7.0 |
| Sterility | Sterile; Non-Pyrogenic |
| In-Use Period | Up to 28 days after first puncture (aseptic handling) |
| Storage | 20–25°C (Controlled Room Temperature) |
| Shelf Life | 24-36 months from manufacture date |
| Intended Use | Research Use Only (RUO) |
| Documentation | Certificate of Analysis provided with each batch |
Premium Bacteriostatic Water with 0.9% Benzyl Alcohol | 30mL Multi-Dose Vial | 48-Hour Delivery Across EU & UK
Bacteriostatic Water: The Essential Diluent for Peptide Research and Reconstitution
eupeptidelap.co.uk is proud to present Bacteriostatic Water for Injection 30mL, a premium sterile solution formulated with 0.9% benzyl alcohol as a bacteriostatic preservative. As a trusted peptide vendor uk and leading EU peptide supplier, we provide researchers across Europe with high-quality bacteriostatic water for sale that delivers exceptional purity, consistency, and documented quality for peptide reconstitution and laboratory applications .
Bacteriostatic Water is a sterile, non-pyrogenic preparation of water for injection containing 0.9% (9 mg/mL) benzyl alcohol added as a bacteriostatic preservative . It is specifically designed to dilute or dissolve research reagents that require an aqueous vehicle before use, making it an indispensable tool in peptide research workflows. The benzyl alcohol preservative inhibits bacterial growth, allowing repeated withdrawals from the same multi-dose vial for up to 28 days under appropriate aseptic handling .
The key advantage of bacteriostatic water over sterile water lies in its multi-dose capability. While sterile water without preservatives must be discarded after a single use, bacteriostatic water’s preservative system enables safe repeated access to the same vial, reducing waste and improving workflow efficiency in research settings . This makes it particularly valuable for peptide research, where multiple doses are often required over extended periods.
For researchers seeking to buy peptide online EU, having high-quality bacteriostatic water is essential for proper peptide reconstitution and storage. Our 30mL multi-dose vials provide the ideal volume for research applications, balancing convenience with economy. Whether your laboratory is based in London, Berlin, Paris, or anywhere in the European Union, our guaranteed 48 hour delivery peptide service ensures your essential supplies arrive when you need them.
The Critical Role of Bacteriostatic Water in Peptide Research
Why Bacteriostatic Water is Essential for Peptide Reconstitution
Most research peptides are supplied as lyophilized (freeze-dried) powders that require reconstitution before use . This critical process demands a sterile, compatible diluent that maintains peptide stability while preventing microbial contamination during multi-dose protocols.
Preservation Against Bacterial Growth: The 0.9% benzyl alcohol in bacteriostatic water acts as a bacteriostatic preservative, inhibiting the growth of bacteria that may be introduced during repeated vial access . This allows researchers to use the same vial for multiple withdrawals over several weeks without compromising sterility.
Extended Multi-Dose Viability: Unlike preservative-free sterile water that must be discarded after a single use, bacteriostatic water supports safe repeated access to the same vial for up to 28 days . This extended viability reduces waste, conserves resources, and improves workflow efficiency in research settings .
Compatibility with Peptides: Bacteriostatic water is widely used as a diluent for peptides and proteins in research workflows, with a broad pH compatibility (4.5–7.0) that suits most research reagents . The formulation maintains the stability and sterility of sensitive substances like peptides and biologic drugs, making it suitable for extended use protocols .
Proper Reconstitution Protocol
Following proper reconstitution procedures is essential for maintaining peptide integrity and ensuring research reproducibility :
-
Disinfect vial tops of both the peptide vial and bacteriostatic water vial with alcohol swabs
-
Draw air into the syringe equal to the amount of bacteriostatic water to be used, then inject this air into the water vial to avoid pressure build-up
-
Withdraw the prescribed amount of bacteriostatic water
-
Inject slowly into the peptide vial, allowing it to run down the inside wall to minimize bubbling and foaming
-
Gently swirl the vial (do not shake) until the powder fully dissolves—shaking can damage peptide bonds
-
Label the vial with the date of reconstitution
-
Refrigerate immediately at 2-8°C
Storage Guidelines for Bacteriostatic Water
Proper storage is critical for maintaining the sterility and effectiveness of bacteriostatic water :
Unopened Vial Storage:
-
Store in a cool, dry place at controlled room temperature (15–25°C / 68–77°F)
-
Protect from direct sunlight and high humidity
-
Avoid exposure to extreme temperatures
-
Shelf life is typically 2-3 years from manufacture date
Opened Vial Storage:
-
Once punctured, use within 28 days per USP guidelines
-
Refrigeration is not required but may be preferred; always follow laboratory protocols
-
Keep vial upright and securely sealed
-
Label with the date of first use
-
Always use sterile syringes and aseptic technique for each withdrawal
Signs of Compromised Solution: Discard the vial immediately if any of the following are observed :
-
Cloudy appearance
-
Visible particles or discoloration
-
Unusual odor
-
Vial open for more than 28 days
-
Past expiration date
Bacteriostatic Water vs. Sterile Water: Key Differences
| Feature | Bacteriostatic Water (0.9% Benzyl Alcohol) | Sterile Water (Preservative-Free) |
|---|---|---|
| Preservative | Contains 0.9% benzyl alcohol | No preservative |
| Multi-Dose Use | Up to 28 days after first puncture | Single use only; discard immediately after opening |
| Waste Reduction | Enables full vial utilization | High waste; unused portion must be discarded |
| Cost Efficiency | More economical for multi-dose protocols | Higher cost per dose for multi-dose applications |
| Primary Application | Peptide reconstitution, multi-dose protocols | Single-dose applications, specific compatibility requirements |
| Storage After Opening | 28-day in-use period with proper aseptic handling | Must be used immediately |
Quality Assurance: Setting the Standard for Research Supplies
Manufacturing Excellence
eupeptidelap.co.uk sources Bacteriostatic Water for Injection 30mL from certified GMP facilities with rigorous quality control protocols:
-
Sterile Manufacturing: Produced under stringent conditions with 0.22 µm filtration and aseptic filling
-
ISO-Certified Facilities: Manufactured in ISO 13485:2016 and GMP-certified facilities
-
Batch-Specific Certificates of Analysis: Complete documentation for each production run
-
Non-Pyrogenic Formulation: Suitable for sensitive research workflows
-
Material Traceability: Full documentation and batch-to-batch consistency
Key Benefits
-
Premium Quality Manufacturing: GMP-certified production, sterile and non-pyrogenic formulation
-
0.9% Benzyl Alcohol Preservative: Inhibits bacterial growth for safe multi-dose use
-
28-Day In-Use Period: Supports repeated withdrawals for up to 28 days with proper aseptic handling
-
30mL Multi-Dose Vial: Convenient size for research applications, reducing waste and improving efficiency
-
Broad pH Compatibility: pH range 4.5–7.0 suitable for most research reagents
-
Peptide Reconstitution: Ideal for dissolving lyophilized peptides and proteins
-
Research-Grade Formulation: Developed for laboratory and research use only (RUO)
-
Reduces Waste: Enables full utilization of vial contents, unlike single-use sterile saline
-
Cost-Effective: More economical for multi-dose research protocols
-
Compatible with Various Research Reagents: Suitable for peptides, proteins, antibodies, and other biologics
-
Extended Multi-Dose Viability: Allows safe repeated access without compromising sample integrity
-
ISO-Certified Manufacturing: Produced in ISO 13485:2016 and GMP-certified facilities
-
Batch-to-Batch Consistency: Rigorous quality control ensures lot-to-lot reproducibility
-
Comprehensive Documentation: Certificates of Analysis with batch-specific data
-
48-Hour EU & UK Delivery: Rapid shipping to research facilities across Europe
-
Easy Integration: Compatible with standard lab handling procedures
-
No Refrigeration Required: Store at room temperature for convenience
-
Safety-Engineered Vial: Standard multi-dose vial compatible with routine laboratory equipment
-
Trusted by Researchers: Positive customer experiences in peptide reconstitution workflows
Frequently Asked Questions
Q: What is Bacteriostatic Water for Injection?
A: Bacteriostatic Water for Injection is a sterile, non-pyrogenic preparation of water for injection containing 0.9% (9 mg/mL) benzyl alcohol as a bacteriostatic preservative . It is designed to dilute or dissolve research reagents that require an aqueous vehicle before use, such as lyophilized peptides .
Q: What is the difference between bacteriostatic water and sterile water?
A: Bacteriostatic water contains 0.9% benzyl alcohol as a preservative, which inhibits bacterial growth and allows multi-dose use for up to 28 days after first puncture . Sterile water contains no preservative and must be discarded after a single use . Bacteriostatic water is more economical and practical for multi-dose research protocols .
Q: How long can I use a vial of bacteriostatic water after first puncture?
A: With proper aseptic handling, bacteriostatic water can be used for up to 28 days after first puncture . After this period, sterility cannot be guaranteed, and the vial should be discarded .
Q: How should I store bacteriostatic water?
A: Store unopened vials at controlled room temperature (20–25°C / 68–77°F) in a cool, dry place away from direct sunlight and high humidity . After opening, continue to store at room temperature and use within 28 days .
Q: What are the signs that bacteriostatic water has gone bad?
A: Discard the vial immediately if you observe any of the following: cloudy appearance, visible particles or discoloration, unusual odor, vial open for more than 28 days, or past expiration date .
Q: Is bacteriostatic water suitable for all peptides?
A: Bacteriostatic water is widely used for peptide reconstitution and is compatible with most peptides . However, always consult the specific peptide’s datasheet for recommended diluent and pH compatibility . Some peptides may have specific requirements that should be verified before use.
Q: Can I use bacteriostatic water for human or animal injection?
A: No. Our bacteriostatic water is strictly for research and laboratory use only (RUO) . It is not for human or animal consumption, and must not be used for therapeutic, diagnostic, or clinical applications. The product has not been tested for use in diagnostics or drug development .
Q: What is the pH range of bacteriostatic water?
A: The typical pH range is 4.5–7.0, which is compatible with most research reagents .
Q: Do you ship bacteriostatic water to EU countries?
A: Yes. As a dedicated EU peptide supplier, we ship Bacteriostatic Water for Injection 30mL to all European Union member states with our guaranteed 48 hour delivery peptide service. Our EU fulfilment centre ensures rapid delivery without customs delays.
Q: What documentation do you provide with bacteriostatic water orders?
A: Every order includes a Certificate of Analysis (COA) with batch-specific purity and sterility data. Additional documentation is available upon request for researchers requiring comprehensive analytical verification.
Q: What is the correct technique for withdrawing bacteriostatic water from a multi-dose vial?
A: Always use aseptic technique for every withdrawal :
-
Disinfect the vial top with an alcohol swab
-
Draw air into the syringe equal to the amount to be withdrawn
-
Inject air into the vial to equalize pressure
-
Withdraw the desired volume
-
Use a fresh sterile needle for each vial access
-
Clean the rubber stopper with an alcohol swab before each puncture
Q: Why is the 28-day rule critical for bacteriostatic water?
A: Even with benzyl alcohol as a preservative, bacteriostatic water can lose effectiveness over time. After 28 days, the preservative may not be strong enough to prevent bacterial growth . Using expired or contaminated water increases the risk of compromised research results and potential contamination of valuable peptides.
Q: Can bacteriostatic water be refrigerated?
A: While refrigeration is not required (storage at 20–25°C is recommended), some researchers prefer to refrigerate for consistency with peptide storage protocols . If refrigerated, allow the solution to reach room temperature before opening to prevent condensation. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
Q: Do you offer bulk quantities of bacteriostatic water for institutional research?
A: Yes. We accommodate bulk orders for research institutions. Contact our team at sales@eupeptidelap.co.uk for volume pricing, custom requirements, and supply agreements for ongoing research programs.
Complete Your Peptide Research Setup with Bacteriostatic Water
eupeptidelap.co.uk is your trusted source for Bacteriostatic Water for Injection 30mL and other essential peptide research supplies. Whether you are reconstituting BPC-157, TB-500, GHK-Cu, or any other research peptide, our high-quality bacteriostatic water provides the sterile, preserved diluent needed for successful research protocols.
The 30mL multi-dose vial format offers the ideal balance of convenience and economy, supporting up to 28 days of repeated withdrawals with proper aseptic technique . Combined with our comprehensive range of research peptides and laboratory accessories, we provide everything you need for advanced peptide research. ...........................................
Order today and experience the eupeptidelap.co.uk difference – premium quality, rapid 48-hour delivery across the EU and UK, and expert support for the European research community.
Related products
Buy Peptide Online EU
Buy Peptide Online EU
Buy Peptide Online EU
Buy Peptide Online EU
Buy Peptide Online EU
Buy Peptide Online EU
Buy Peptide Online EU










Buy Peptide Online EU
Thymosin Alpha 1 5mg